We’re a full-stack drug discovery and development company for inherited metabolic diseases: congenital disorders of glycosylation, mitochondrial disorders, GPI-anchor defects, tRNA-synthetase syndromes, and the long tail of ultra-rare metabolic biology.

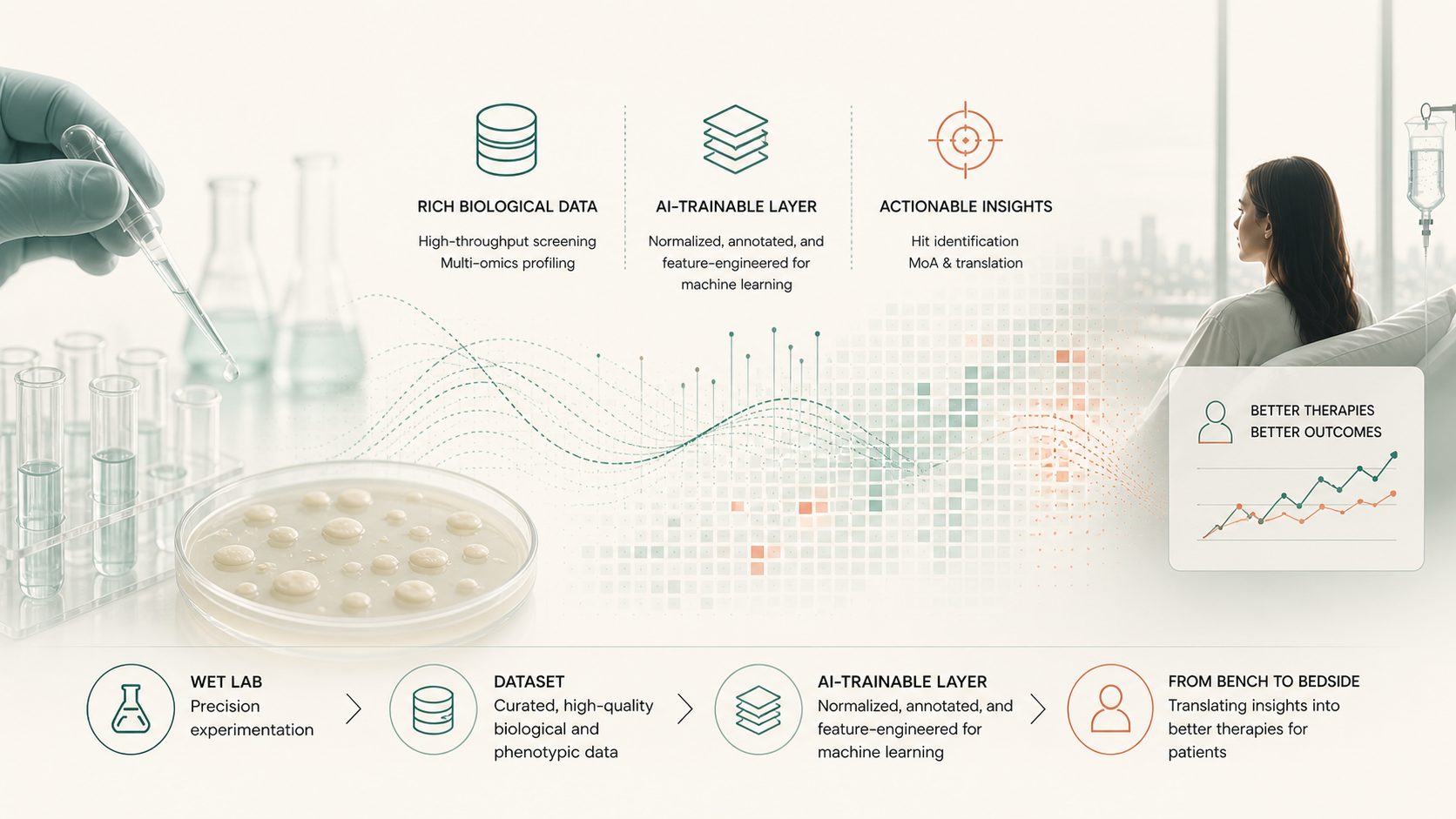
Inherited metabolic diseases (IMDs) are individually ultra-rare but collectively enormous — about 15% of the world’s ~10,000 rare diseases. Most have a single known causitive gene, a pediatric onset, and no approved therapy. They’re exactly the programs legacy pharma de-prioritizes, and exactly where a yeast-first, patient-partnered approach excels.
Our explicit goal: scale from a dozen active IMD programs today to several hundred over the next decade. That's a sizeable chunk of the addressable IMD universe, and a transformative expansion over the status quo.
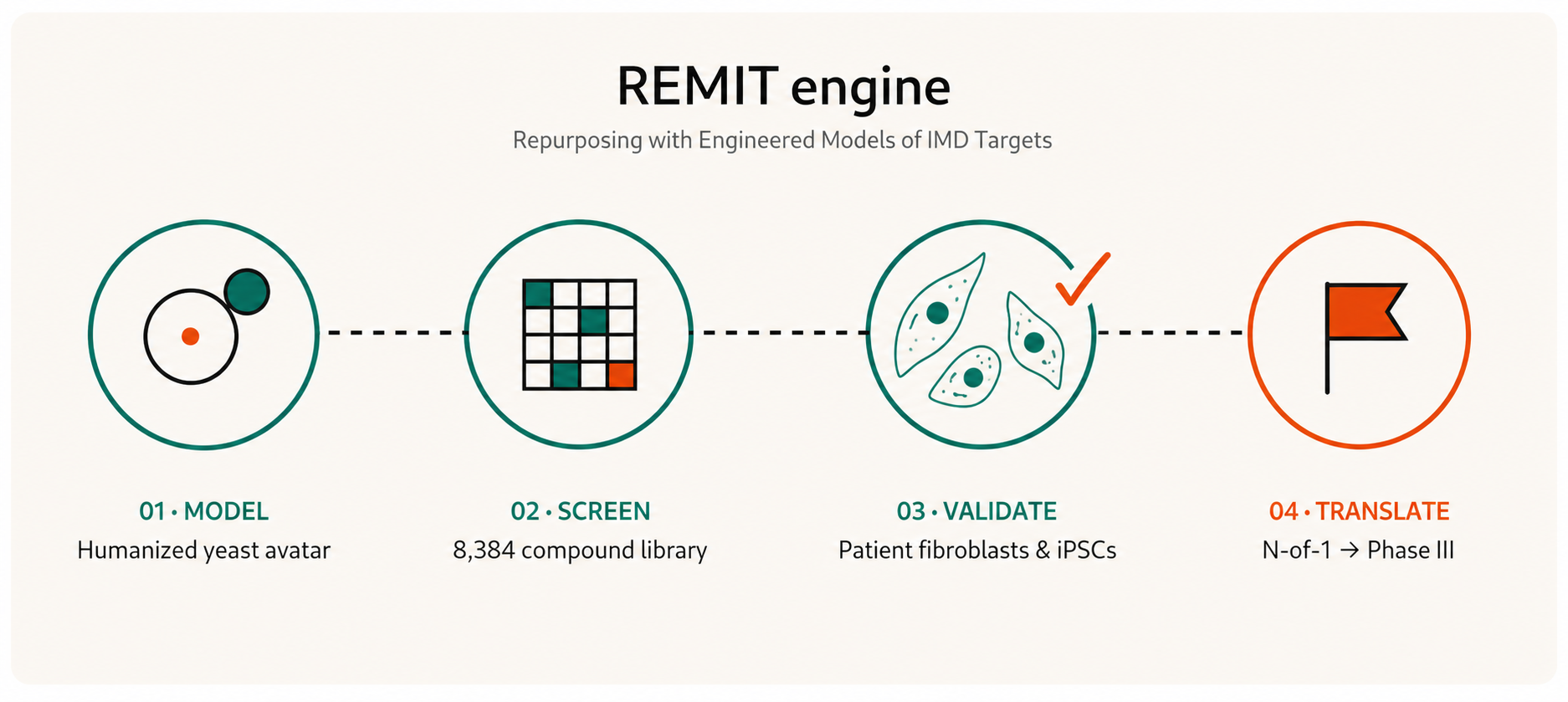
Pharmacological deficiency models of every electron transport chain complex (CI–CV), N-glycosylation defects (PMM2, ALG11, DHDDS), GPI-anchor disorders (PIGN, PIGW), and aminoacyl-tRNA synthetases (NARS1, FARS2, MTAARS2).
Every program is anchored by a specific family or foundation. The yeast screen, the patient-cell validation, the 1-to-N protocol, and the registrational trial are all on one shared timeline.
A parent-initiated N-of-1 study of a decades-old Japanese diabetic-neuropathy drug became a 38-patient Phase III at Mayo Clinic Minnesota in under three years for less than $5M. That cadence is the Perlara 3.0 standard.
“We went from a single child on compassionate-use epalrestat to a registrational trial in three years on less than $5M in patient-foundation capital. This is what community medicine looks like.”
Repurposing with Engineered Models of Inherited metabolic disease Targets. A four-stage engine: build the yeast avatar, screen a standardized library, validate in patient cells, advance through IND-enabling and trials.
Our working notebook is Perlara’s Substack, Cure Odysseys, where each Cure Odyssey is a single disease, family, and yeast model moving together through the pipeline.
Six of twelve top yeast rescuers for SURF1 Leigh Syndrome reproduced in patient fibroblasts — the strongest yeast-to- human translation signal we’ve measured.
In GMPPA-CDG, loss of GMPPA causes runaway GMPPB and hypermannosylation. Two wrongs make a right: dosing a GMPPB inhibitor restores flux.
How a Japanese aldose-reductase inhibitor approved for diabetic neuropathy became a glycosylation-rescue drug candidate on its way to a Phase 3 study.
All Cure Odysseys posts →